Tungsten
W • Atomic Number 74
Tungsten
Tungsten is a shiny white, exceptionally hard, very dense, and rare metal with good electrical conductivity. At 3,422 degrees Celsius, it has the highest melting point of all chemical elements.
Its best-known use is as a filament in light bulbs.
The most important application is as an alloy additive in steels to increase hardness and strength in industrial tools, as well as tungsten carbide in industrial tools and military applications.
China is by far the largest producer of tungsten, accounting for over 83% of global production.
The Xihuashan Mine in China is the largest active tungsten mine in the world. It is operated by China Tungsten High-Tech.
Due to its rarity and military applications, tungsten is considered a critical raw material in the EU, the US, Japan, South Korea, the UK, Australia, and Canada.
-
History
In 1781, Swedish chemist Carl Wilhelm Scheele discovered tungstic acid in a mineral now known as scheelite. His compatriot Torbern Bergman concluded that a new metal could be produced from the acid.
The brothers and chemists Juan José and Fausto Elhuyar succeeded in doing so in 1783 by reducing wolframite from the Saxon Ore Mountains with charcoal.The name tungsten goes back to the Freiberg mineralogist Georgius Agricola, who probably described a tungsten-containing mineral as early as the 16th century and gave it the Latin name “lupi spuma,” which means wolf foam. The term “tungsten,” commonly used in other languages, comes from Swedish and means “heavy stone.”
Due to its extremely high melting point, tungsten was initially difficult to process.
From the mid-19th century onwards, tungsten was used in steel alloys to manufacture harder tools.
At the end of the 19th century, with the advent of electric light, tungsten wire, which did not melt as quickly, replaced carbon filaments in light bulbs.
In the 20th century, tungsten carbide (WC) was developed for armor-piercing ammunition and extremely hard cutting tools.
-
Applications and Uses
Due to its exceptional physical properties, tungsten is used in a wide range of applications. Tungsten carbide is used in the manufacture of hard metals for cutting, mining, and wire drawing tools, as well as in the defense industry for armor-piercing ammunition, grenades, and armor plating.
In alloys, tungsten provides heat resistance in valves, turbine wheels of aircraft engines, and rocket nozzles, while also protecting against wear.
Tungsten is also used in electronics in electrodes, in the semiconductor industry for microchips, and in X-ray tubes.
Due to its density, tungsten is also used for radiation shielding.
-
Occurrence, Mining and Extraction
Important ores include wolframite and scheelite.
Over 80 percent of tungsten is mined in China, which also has some of the largest tungsten deposits. The Xihuashan Mine in Jiangxi Province is the largest active tungsten mine in the world. It is operated by Tungsten High-Tech, a subsidiary of the state-owned China Minmetals Corporation.
After China, Vietnam is the second largest mining country for tungsten. The Nui Phao Mine in Vietnam is the largest tungsten mine outside China. It is operated by Masan Resources. Russia ranks third, but figures on production volumes are scarce.
There are also several tungsten producers in the EU. In Austria, Wolfram Bergbau und Hütten AG operates a small underground mine in the Alps. The company, which belongs to the Swedish Sandvik Group, obtains most of its raw material from recycling. In addition, there are smaller tungsten mines in operation in Portugal and Spain.
Global annual production amounts to around 80,000 tons.
The recycling of scrap is an important source for the tungsten industry.
-
Substitution
Possible substitutes for tungsten carbides are hard metals based on molybdenum carbide, niobium carbide, or titanium carbide, ceramics, ceramic-metal composites, and tool steels. Most of these options reduce tungsten consumption rather than replacing it.
Possible substitutes for other applications include: molybdenum for certain tungsten rolled products; molybdenum steels for tungsten steels, although most molybdenum steels still contain tungsten.
Depleted uranium or lead replace tungsten or tungsten alloys in applications requiring high density or radiation shielding capabilities.
Depleted uranium alloys or hardened steel can substitute for tungsten carbides or tungsten alloys in armor-piercing projectiles.
In some applications, substitution would result in higher costs or compromised performance. -
Unique Properties
Physical Properties
Tungsten is a shiny white metal that is ductile in its pure state and has high hardness, density, and strength. Its density is almost as high as that of gold, its Brinell hardness is 250 HB, and its tensile strength ranges from 550-620 N/mm² (soft) to 1920 N/mm² (hard). The metal exists in a stable cubic body-centered α modification with a lattice constant of 316 pm at room temperature. This type of crystal structure is often referred to as the tungsten type. A substance known as a metastable β modification of tungsten (distorted body-centered cubic) is, however, the tungsten-rich oxide W3O.
After carbon, tungsten has the second highest melting point of all chemical elements at 3422 °C. Its boiling point of 5555 °C is surpassed only by the rare metal rhenium at 5596 °C, which is 41 K higher.
The metal is a superconductor with a transition temperature of 15 mK.
.Chemical Properties
Tungsten is a chemically very resistant metal that is hardly attacked even by hydrofluoric acid and aqua regia (at least at room temperature). However, it dissolves in mixtures of hydrochloric and nitric acid and molten mixtures of alkali nitrates and carbonates.
-
Isotopes
A total of 33 isotopes and 5 nuclear isomers of tungsten are known. Of these, five isotopes — ¹⁸⁰W, ¹⁸²W, ¹⁸³W, ¹⁸⁴W, and ¹⁸⁶W — occur naturally. Among them, the isotope ¹⁸⁴W has the highest natural abundance.
For a long time, all five natural isotopes were considered stable. However, in 2004, the CRESST experiment at the Laboratori Nazionali del Gran Sasso succeeded, as a byproduct of its search for dark matter, in detecting that the isotope ¹⁸⁰W undergoes alpha decay. Its half-life is extremely long — about 1.8 trillion years — making this decay undetectable under normal laboratory conditions.
The radioactivity of this natural isotope is so low that it can be ignored for all practical purposes.
In contrast, the artificial radioactive isotopes of tungsten have much shorter half-lives, ranging from 0.9 millisecondsfor ¹⁸⁵W to 121.2 days for ¹⁸¹W. -
Safety Information
As a powder or dust, it is easily flammable; in compact form, it is non-combustible.
-
Compounds
Oxides
Tungsten forms several oxides. Between the initial compound:
- Tungsten(VI) oxide WO₃ – lemon yellow
and the final compound:
- Tungsten(IV) oxide WO₂ – brown
there are the following intermediate oxides:
-
W₁₀O₂₉, blue-violet, homogeneity range WO₂.₉₂–WO₂.₈₈
-
W₄O₁₁, red-violet, homogeneity range WO₂.₇₆–WO₂.₇₃
-
W₁₈O₄₉, WO₂.₇₂
-
W₂₀O₅₀, WO₂.₅₀
Other Compounds
-
Sodium tungstate Na₂WO₄
-
Zirconium tungstate ZrW₂O₈ – shows an anomaly upon heating.
-
Tungsten bronzes MₓWO₃, where M = alkali, alkaline earth, or lanthanoid metal, ca. 0.3 < x < 0.9. These are electrically conductive and exhibit intense colors depending on metal content.
-
Calcium tungstate CaWO₄, known as the mineral scheelite.
-
Tungsten carbide WC, an extremely hard, metallic-like compound. There is also ditungsten carbide W₂C.
-
Tungsten hexafluoride WF₆
-
Lead tungstate PbWO₄
-
Tungsten disulfide WS₂, used as a dry lubricant (similar to MoS₂).
Uses of the Compounds
Tungsten carbide is used as a neutron reflector in nuclear weapons to reduce the critical mass. Tungsten carbides (hard metals) are also used in machining due to their high hardness.
Tungstates are used to impregnate fabrics, making them flame-retardant.
Tungsten-based pigments are used in painting, as well as in the ceramics and porcelain industries.
Lead tungstate is used as a modern scintillator in particle physics.
General Information Name, Symbol, Atomic Number Tungsten, W, 74 Series Transition metals Group, Period, Block 6, 6, d Appearance Grayish-white, shiny CAS Number 7440-33-7 Proportion in Earth’s crust 64 ppm Atomic Properties Atomic mass 183,84 u Atomic radius (calculated) 135 (193) pm Covalent radius 162 pm Electron configuration [Xe] 4f145d46s2 1. ionization energy 770 kJ/mol 2. ionization energy 1700 kJ/mol Physical Properties State of matter Solid Crystal structure Body-centered cubic Density 19,3 g/cm3 (20 °C) Mohs hardness 7,5 Magnetism Paramagnetic (  = 7,8 · 10−5)
= 7,8 · 10−5)Melting point 3695 K (3422 °C) Boiling point 5828 K (5555 °C) Molar volume 9,47 · 10−6 m3/mol Heat of vaporization 824 kJ/mol Heat of fusion 35,4 kJ/mol Speed of sound 5174 m/s Specific heat capacity 138 J/(kg · K) Electrical conductivity 18,52 · 106 A/(V · m) Thermal conductivity 170 W/(m · K) Chemical Properties Oxidation states 6, 5, 4, 3, 2 Standard electrode potential −0,119 V (WO2 + 4H+ + 4e−
→ W + 2H2O)Electronegativity (Pauling) 2,36 Isotopes Isotop NH t1/2 ZA ZE (MeV) ZP 178W {syn.} 21,6 d ε 0,091 178Ta 179W {syn.} 37,05 min ε 1,060 179Ta 180W 0,13 % 1,8 · 1018 a α 2,516 176Hf 181W {syn.} 121,2 d ε 0,188 181Ta 182W 26,3 % Stable 183W 14,3 % Stable 184W 30,67 % Stable 185W {syn.} 75,1 d β− 0,433 185Re 186W 28,6 % Stable 187W {syn.} 23,72 h β− 1,311 187Re 188W {syn.} 69,4 d β− 0,349 188Re NMR Properties Isotope Spin γ
(rad·T−1·s−1)Er(1H) fL (MHz) at
B = 4,7 T183W 1/2 1,128 · 107 1,07 · 10−5 4,166 Safety Information GHS Hazard Identification (powder form) 
- H- and P-statements: H: 228 EUH: none P:210–240–241–280–370+378
Hazard Labeling

Highly flammable (F) Powder — R and S phrases: R: 11 S: 43

- Hits: 747


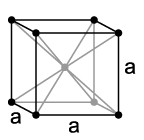
 and the lattice parameter a = 302.4 pm, as well as two formula units per unit cell.
and the lattice parameter a = 302.4 pm, as well as two formula units per unit cell.