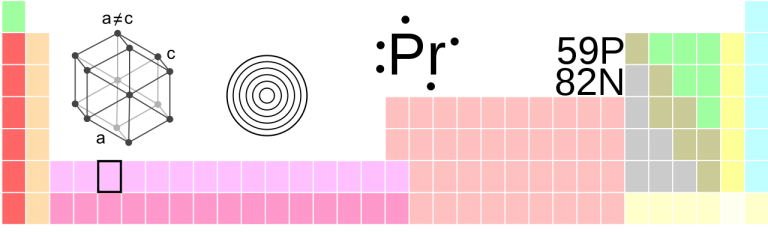
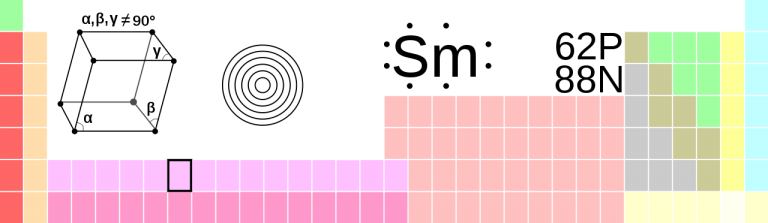
Samarium
Sm • Atomic Number 62

Samarium
Samarium is a silvery-white, shiny metal that is relatively hard and brittle. It is neither malleable nor ductile. Samarium belongs to the group of middle rare earth elements.
Its key characteristic is that it is paramagnetic at room temperature—it is attracted by an external magnetic field but does not retain permanent magnetism on its own. However, its alloys, particularly with cobalt, are among the strongest permanent magnetic materials known.
By far the most significant and economically important application of samarium is its use in samarium–cobalt (SmCo) magnets.
In 1839, Swiss chemist Jean Charles Galissard de Marignac analyzed a mineral from the Russian Ural Mountains. He discovered a new oxide in the mineral, which he called “yttrium earth.” While he suspected it contained a new element, he could not clearly identify it.
In 1879, French chemist Paul-Émile Lecoq de Boisbaudran worked with the so-called "didymium earth," which at the time was believed to be a single element. Through extensive fractional crystallization, he succeeded in observing a new, specific spectral line in the visible range, indicating the presence of a distinct element.
Shortly after Lecoq de Boisbaudran, Swedish chemist Lars Fredrik Nilson isolated an oxide from the same mineral, samarskite, which he believed to be identical to Lecoq de Boisbaudran’s samarium.
Later, both De Marignac and Lecoq de Boisbaudran confirmed that Nilson’s sample was in fact a mixture of samariumand a new element, which was later named gadolinium.
The name of the mineral and the element samarium derive from Colonel Samarsky, a Russian mining official who first discovered the mineral.
In 1903, German chemist Wilhelm Muthmann succeeded in producing metallic samarium via electrolysis.
-
Occurence
The two most economically significant sources for the extraction of samarium are monazite and bastnäsite.
The phosphate mineral monazite contains a higher concentration of samarium—around 1 to 3 percent—compared to bastnäsite. However, bastnäsite deposits are generally preferred, as they contain significantly lower levels of radioactive thorium, making processing and handling safer and less regulated.
China is by far the world's largest producer of samarium.
-
Extraction
Starting from monazite or bastnäsite, the separation of rare earth elements is carried out using ion exchange, solvent extraction, or electrochemical deposition. In a final processing step, high-purity samarium oxide is reduced to the metal using metallic lanthanum and subsequently purified by sublimation.
-
Application
The most important and economically significant application of samarium is the production of samarium-cobalt magnets (SmCo magnets). These are among the strongest permanent magnets in the world and are indispensable for many high-tech applications.

Samarium-Cobalt Magnet
SmCo magnets are extremely resistant to demagnetization and remain stable even when exposed to strong external magnetic fields.
Their greatest advantage is their high thermal stability: they retain their magnetic properties at elevated temperatures up to 350 °C—and even beyond. This makes them unrivaled in high-temperature applications.
Unlike NdFeB magnets, SmCo magnets are corrosion-resistant and typically do not require any protective coating.
Key applications for SmCo magnets include aerospace, military and defense technologies (e.g., radar systems, guidance and control systems), high-performance motors, and precision instruments such as traveling wave tubes (TWTs) used in radar and satellite communication.
Additionally, samarium is used in neutron absorbers in nuclear reactors and as a catalyst in the chemical industry.

- Hits: 565