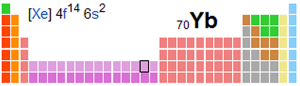
Ytterbium
Yb • Atomic Number 70

Ytterbium
Ytterbium is a soft, silvery-white, malleable metal with unique optical and electronic properties. It belongs to the heavy rare earth elements.
Ytterbium ions have well-defined absorption and emission bands in the near-infrared region (around 980 nm excitation and 1030 nm emission). This property forms the basis for its most important application in ytterbium-doped lasers used in fiber optic networks.
Ytterbium stands out from other elements because it is stable in both the +3 and +2 oxidation states. This “divalency” enables unique chemical reactions.
Another notable property is that ytterbium compounds are often paramagnetic (attracted by magnetic fields). This makes it useful in alloys where magnetic interference is undesirable.
Ytterbium was discovered in 1878 by the Swiss chemist Jean Charles Galissard de Marignac. He studied gadolinite more closely and identified a previously unknown element, which he named ytterbium after the Swedish locality Ytterby, where the gadolinite was found, and due to its similarity to yttrium.
-
Occurence
The main sources for ytterbium extraction are bastnäsite and monazite deposits. These are primarily mined in China, with smaller-scale mining also occurring in the USA, Australia, and Southeast Asia.
The demand for ytterbium is growing due to its use in a variety of technologies.
Ytterbium production is almost exclusively concentrated in China.
-
Extraction
Ytterbium is obtained through solvent extraction. For this process, the rare earth oxide (REO) mixture is dissolved in hydrochloric acid, and the solution is pumped through a series of mixer-settler units. At each stage, an organic solvent is added that selectively binds a specific rare earth element.
Due to slight differences in the acidity of the ions, the various elements move through the cascade at different rates.
Ytterbium accumulates in the later stages of the extraction cascade.
The result of this step is highly pure ytterbium(III) oxide (Yb₂O₃).
To produce metallic ytterbium, the oxide is usually reduced metallothermically using lanthanum or cerium.
The metal obtained by reduction is often not pure enough for high-tech applications. The most important purification method is in-situ distillation.
-
Application
One of the key drivers for ytterbium demand is the expansion of fiber optic networks and 5G. Ytterbium plays a crucial role in high-performance semiconductors and ytterbium-doped fiber amplifiers (YDFAs). Ytterbium-based amplifiers, an essential component in fiber lasers, enable efficient high-speed data transmission in communication technologies.
Another important application of ytterbium is in medical technologies. Ytterbium isotopes are used in medical imaging, radiation therapy, and surgical lasers.
Ytterbium demand could further increase in the future due to innovations in emerging fields such as quantum computing and ultra-precise atomic clocks.
General Information Name, Symbol, Atomic Number Ytterbium, Yb, 70 Series Lanthanoid Groupe, Periode, Block La, 6, f Appearance silvery-white CAS-Number 7440-64-4 Abundance in Earth's crust 2.5ppm Atomic Properties Atomic Mass 173.04 u Atomic Radius 175 pm Covalent Radius 187 pm Electron Configuration [Xe] 4f¹⁴ 6s² 1. Ionization Energy 603.4 kJ/mol 2. Ionization Energy 1174.8 kJ/mol 3. Ionization Energy 2417 kJ/mol 4. Ionization Energy - Physical Properties State of Matter solid Crystal Structure Cubic face-centered Density 6.973 g/cm³ (at 25 °C) Magnetism Paramagnetic (χm = 3.4 * 10⁻⁵)
Melting Point 1097 K (824 °C) Boiling Point 1469 K (1196 °C) Molar Volume 24.84 * 10⁻⁶ m³/mol Heat of Vaporization 160 kJ/mol Heat of Fusion 7.7 kJ/mol Electrical Conductivity 4.0 * 10⁶ A/(V·m) Thermal Conductivity 39 W/(m*K)

- Hits: 581